Since the beginning of the major health crisis stemming from the change of formula of LEVOTHYROX, MERCK, the ANSM and the Minister of Health have all given the same speech: in February 2012, following an official investigation of pharmacovigilance, and to limit differences in the content of active substance according to the batches of the specialty, the French Agency for the Safety of Health Products has asked Merck Santé, license holder of the marketing of Levothyrox specialties, to restrict its Levothyroxine Sodium content specifications within 95/105% of the declared dose over the shelf-life of the product. For that purpose, the company has sought a change in the formula of its specialties, consisting of modifying the excipients used, which was authorised on 27 September 2016 by the National Agency for the Safety of Medicines and Health Products, given the two pharmacokinetic studies demonstrating the bioequivalence of old and new formulations.
As a matter of fact, the official speech is to recall that until 2017, the old formula of LEVOTHYROX comprised from 90 to 111% of active ingredient, which would have been a source of instability of the product.
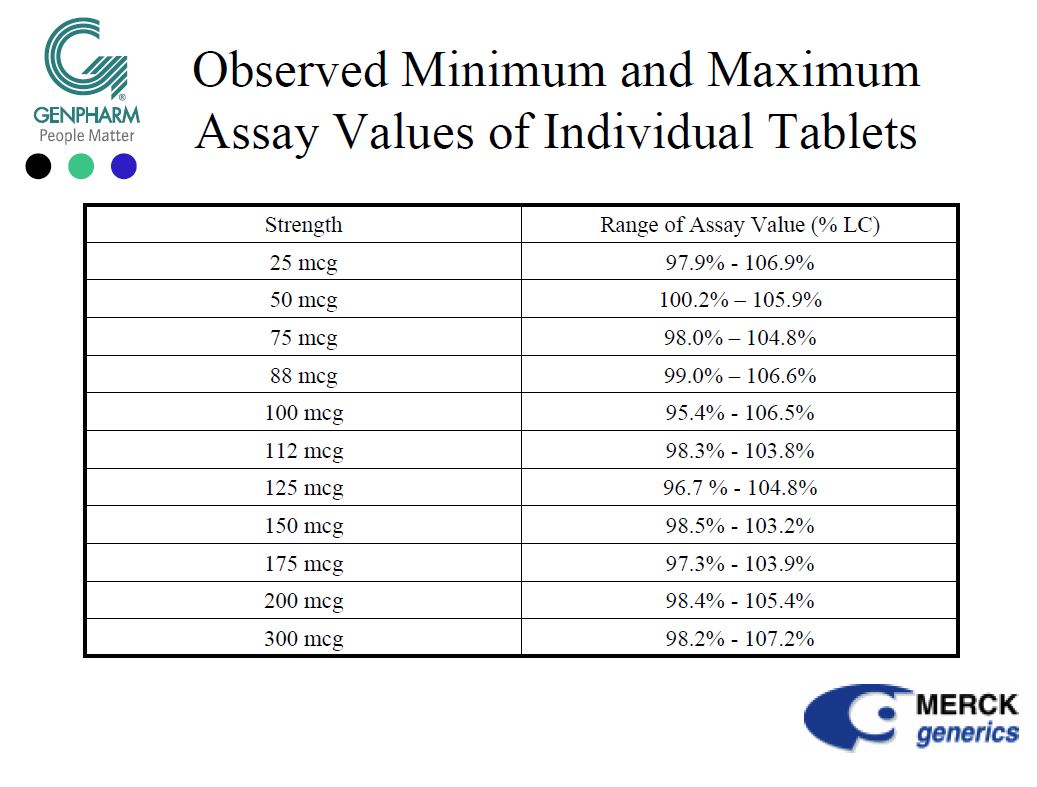
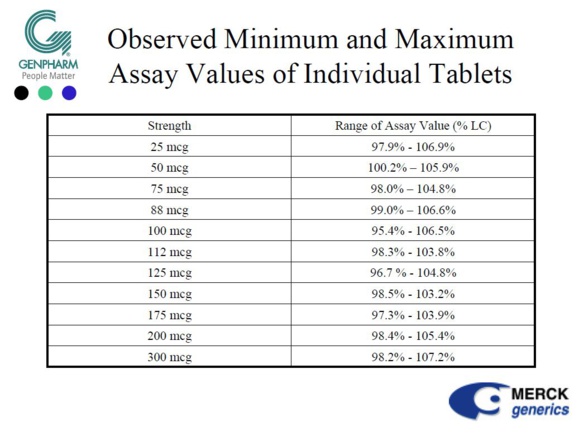
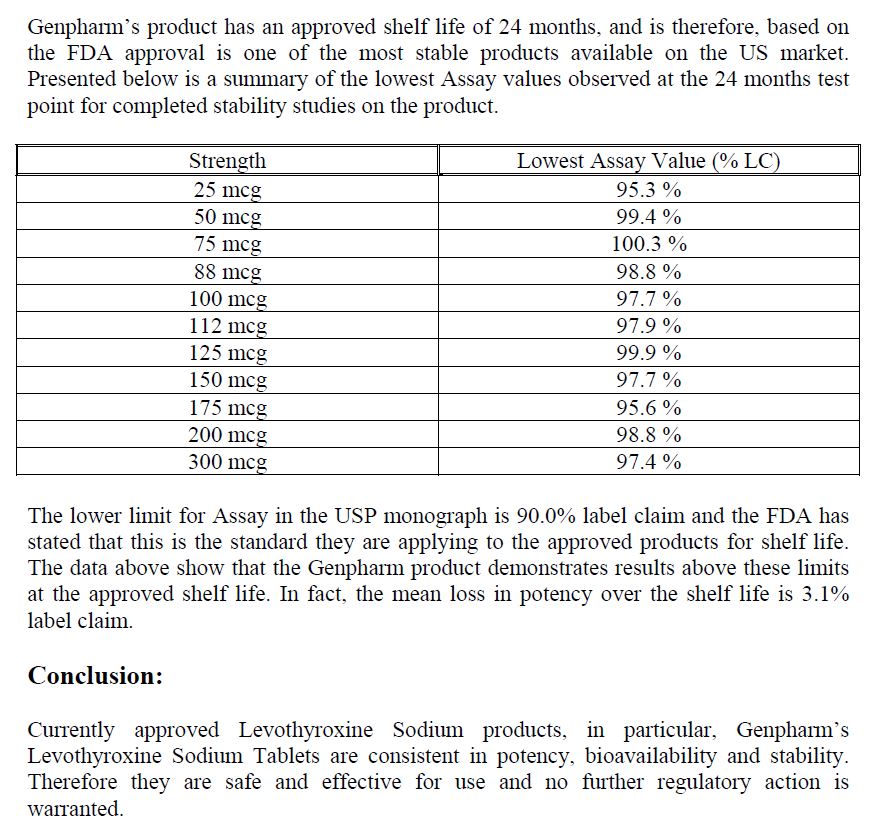
Nevertheless, in 2005, MERCK and its North American subsidiary (GENPHARM) explained to the powerful FDA (Food & Drug Administration) that the levothyroxine sodium marketed in the USA under the name NOVOTHYROX was manufactured in Germany and was not distinguishable from products marketed under other names, apart from the addition of a food colouring in the tablets.
On the other hand, MERCK and GENPHARM boasted that the NOVOTHYROX met the 95/105% standard of active ingredient.
From an industrial point of view, it seems difficult to accept that a single plant produces the same drug with different manufacturing processes affecting the content of active ingredient, especially when the quantities are difficult to measure since we are dealing in microgram (one millionth of a gram).
We must remain cautious as all the constituents of this HEALTH SCANDAL are now put together. Mrs BUZYN should come down from her ivory tower to meet and discuss with the group of sick people affected by this scandal in order to make the only necessary decision i.e. requisitioning the production and distribution of LEVOTHYROX with lactose.
As a matter of fact, the official speech is to recall that until 2017, the old formula of LEVOTHYROX comprised from 90 to 111% of active ingredient, which would have been a source of instability of the product.
Nevertheless, in 2005, MERCK and its North American subsidiary (GENPHARM) explained to the powerful FDA (Food & Drug Administration) that the levothyroxine sodium marketed in the USA under the name NOVOTHYROX was manufactured in Germany and was not distinguishable from products marketed under other names, apart from the addition of a food colouring in the tablets.
On the other hand, MERCK and GENPHARM boasted that the NOVOTHYROX met the 95/105% standard of active ingredient.
From an industrial point of view, it seems difficult to accept that a single plant produces the same drug with different manufacturing processes affecting the content of active ingredient, especially when the quantities are difficult to measure since we are dealing in microgram (one millionth of a gram).
We must remain cautious as all the constituents of this HEALTH SCANDAL are now put together. Mrs BUZYN should come down from her ivory tower to meet and discuss with the group of sick people affected by this scandal in order to make the only necessary decision i.e. requisitioning the production and distribution of LEVOTHYROX with lactose.


 Un avocat ?
Un avocat ?